Experts from Arizona State University of Cambridge (UK) and other research institutes have collaborated closely with AstraZeneca to examine vaccine-induced immune thrombotic thrombocytopenia (VITT syndrome). The researchers’ next goal is to tweak the adenovirus.
An international scientific team has identified a probable explanation for the uncommon blood clots that have developed in individuals who have received an adenovirus vaccine against Covid-19 infection, such as AstraZeneca.
Cardiff University and Arizona State University researchers collaborated with AstraZeneca to evaluate vaccine-induced immune thrombotic thrombocytopenia (VITT), also known as thrombosis with thrombocytopenia syndrome (TTS), a potentially fatal condition that occurs in a very small number of people following vaccination with the Oxford-AstraZeneca or Johnson & Johnson vaccines.
The international team employed cutting-edge technology to analyze the AstraZeneca vaccines in minute detail in order to determine whether the extremely unusual side effect was caused by the viral vector.
Their findings suggest that the viral vector – in this case an adenovirus employed to deliver the coronavirus’ genetic material into cells – and the manner it binds to platelet factor 4 (PF4) following injection could be the mechanism.
The authors speculate that in extremely rare situations, the viral vector may penetrate the bloodstream and attach to PF4, prompting the immune system to recognize the complex as foreign. They hypothesize that this faulty immunity may result in the generation of antibodies against PF4, which bind to and activate platelets, clustering them together and generating blood clots in a very small proportion of individuals following vaccination.
“VITT only happens in extremely rare cases because a chain of complex events needs to take place to trigger this ultra-rare side effect. Our data confirms PF4 can bind to adenoviruses, an important step in unravelling the mechanism underlying VITT,” said Professor Alan Parker.
“Although very rare, it is critical we fully investigate vector-host interactions of the vaccine at a mechanistic level to help us understand both how the vaccine generates immunity – and how it may lead to any rare adverse events, such as VITT. Establishing a mechanism could help to prevent and treat this disorder.
“We hope our findings can be used to better understand the rare side effects of these new vaccines – and potentially to design new and improved vaccines to turn the tide on this global pandemic.”
Both the AstraZeneca and Johnson & Johnson vaccines employ an adenovirus to deliver coronavirus spike proteins into humans and elicit a protective immune response.
When both vaccines developed the extremely unusual side effect VITT, scientists wondered if the viral vector played a role. Another significant signal was that neither the Moderna nor Pfizer vaccines, which were created using an entirely different technology known as mRNA vaccines, demonstrated this effect.
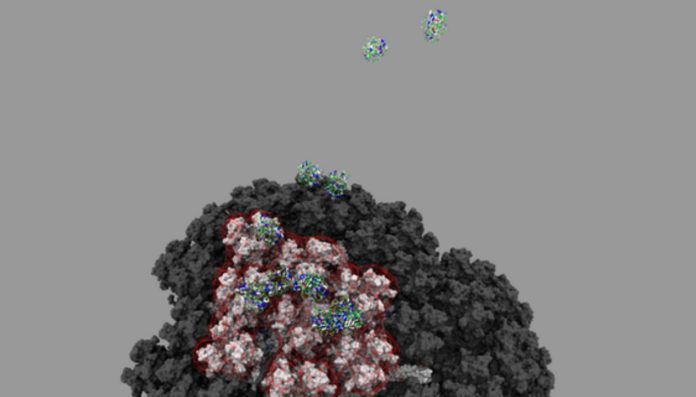
The researcher applied a technique called CryoEM to flash freeze preparations of ChAdOx1, the adenovirus used in the AstraZeneca vaccine, and then bombarded them with electrons to create tiny images of the vaccine components.
They were then able to examine the structure of the virus’s exterior protein cage – the viral capsid – and other essential proteins that allow the virus to enter the cell at an atomic level.
The team describes in detail the structure and receptor of ChAdOx1, which was derived from chimp adenovirus Y25 – as well as how it interacts with PF4. They believe that it is this unique interaction – and the manner in which it is presented to the immune system – that could cause the body’s own defenses to perceive it as foreign, resulting in the production of antibodies against this self-protein.
The research team used computational tools, a specialty of ASU’s Singharoy group, to demonstrate that one of the ways the two molecules bind strongly is through electrostatic interactions.
“We found that ChAdOx1 has a strong negative charge. This means the viral vector can act like a magnet and attract proteins with the opposite, positive charge, like PF4,” said Dr Alexander Baker, first author.
“We then found that PF4 is just the right size and shape that when it gets close to ChAdOx1 it could bind in between the negatively charged parts of ChAdOx1’s surface, called hexons.”
The researchers anticipate that by gaining a better knowledge of what may be generating rare VITT, they will be able to provide additional insight into how vaccines and other therapies that rely on the same technology might be altered in the development of next generation vaccines and medicines.
“With a better understanding of the mechanism by which PF4 and adenoviruses interact there is an opportunity to engineer the capsid, or outer shell of the vaccine, to prevent this interaction occurring. Modifying ChAdOx1 to reduce electronegativity may reduce the chance of causing thrombosis with thrombocytopenia syndrome,” said Dr. Baker.
Source: ScienceAdvances
You were reading: Adenovirus used in AstraZeneca vaccine may be responsible for thrombosis, researchers say