Late-onset retinal degeneration (L-ORD) is a very rare, dominantly inherited illness in which a defective gene from one parent causes the disease. A mutation in the gene that codes for the protein CTRP5 indues L-ORD.
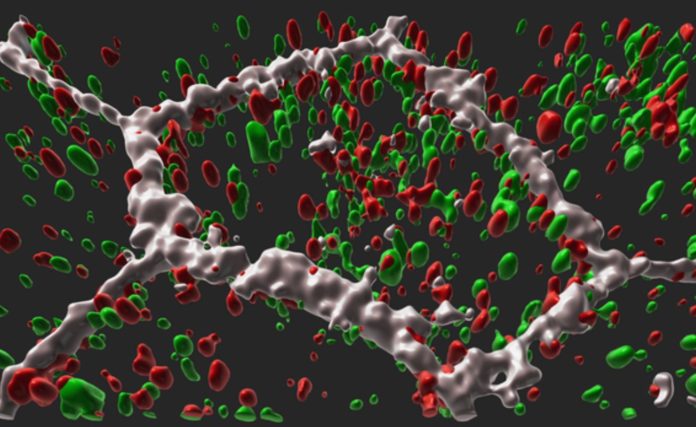
Individuals with the condition have irregular blood vessel growth as well as deposits of apolipoprotein E, which is involved in fat metabolism within the retina. Symptoms such as trouble seeing in the dark and loss of central vision commonly emerge between the ages of 50 and 60. Cells in the retinal pigment epithelium (RPE), a layer of tissue that nourishes the retina’s light-sensing photoreceptors, atrophy and die as L-ORD develops. The loss of RPE results in the loss of photoreceptors and, as a result, the loss of eyesight.
A group of researchers has discovered that gene therapy and the diabetes drug metformin could be possible treatments for late-onset retinal degeneration (L-ORD), a rare and disabling eye disease. To study the disease, researchers from the National Eye Institute (NEI), which is part of the National Institutes of Health, created a “disease-in-a-dish” model.
“This new model of a rare eye disease is a terrific example of translational research, where collaboration among clinical and laboratory researchers advances knowledge not by small steps, but by leaps and bounds,” says Michael F. Chiang, M.D., director of the NEI, part of the National Institutes of Health.
The team led by Dr. Kapil Bharti created a laboratory model that uses induced pluripotent stem cells (fibroblasts) derived from skin to produce RPE. They created RPE from two siblings who had L-ORD and compared it to RPE from two of the patients’ unaffected siblings who did not have the disease-causing mutation.
Apolipoprotein E deposits around the tissue and aberrant discharges of vascular endothelial growth factor, a protein that drives blood vessel formation, were found in the patient-derived RPE, which shared essential characteristics with the condition in humans. The RPE cells were likewise deformed or dysmorphic. The RPE of the unaffected siblings, on the other hand, appeared to be normal. The researchers also discovered that as compared to the models created from unaffected siblings, the patient-derived RPE secreted substantially less of the mutant and non-mutant CTRP5 protein.
They demonstrated that mutant CTRP5 was less likely to connect with cell receptors that assist fine-tune fat metabolism control using a computer modeling technique. Less receptor binding in turn leads to chronic activation of AMP-activated protein kinase (AMPK), a key regulator of energy homeostasis and fat metabolism.
They hypothesized that persistently active AMPK becomes less sensitive to energy demand and food supply mismatches. When metabolic abnormalities go untreated, they disrupt lipid metabolism, which explains why apolipoprotein E accumulates near the RPE layer as deposits.
Chemical inhibition of chronically active AMPK in the patient-derived RPE model resulted in less deposits of apolipoprotein E and less abnormal release of vascular endothelial growth factor, according to the researchers.
They then examined two potential therapeutic techniques using the patient-derived RPE model: a gene therapy approach to induce normal CTRP5 expression in the RPE model, and the diabetes drug metformin, which appears to alter AMPK activity, re-sensitizing it to changes in cellular energy status. In RPE models, both techniques inhibited the appearance of L-ORD.
“Importantly, we now have two potential strategies to disrupt the L-ORD disease process. While gene therapy may be years away, metformin is a drug that’s long been used to treat diabetes,” says Bharti, who with NEI collaborators is planning a clinical trial to test the drug in people with L-ORD.
Despite its rarity, L-ORD illness is similar to other retinal degenerations such as age-related macular degeneration, which is a prominent cause of vision loss. The model created for this study could be useful in understanding pathological changes in the RPE as people get older.
Source: 10.1038/s42003-021-02872-x
Image Credit: Ruchi Sharma and Davide Ortolan (NEI)
You were reading: Scientists find a potential treatment for late-onset retinal degeneration