What was the source of energy for the earliest chemical processes at the dawn of life, and how did they get started?
The metabolism of LUCA, the last universal common ancestor, has been rebuilt by researchers at Heinrich Heine University (HHU) Düsseldorf.
They discovered that almost all of the chemical reactions needed by primordial life to put assemble the molecular building blocks of cells release energy. This revealed the long-sought source of energy needed to accelerate these processes, which had been lurking in plain sight.
As long as one vital beginning chemical is present, the energy required to generate the building blocks of life originates from within metabolism. The cleanest, greenest, newest, and oldest of all energy carriers, hydrogen gas, H2, is the hidden component that releases energy from within at life’s origin.
Prof. Dr. William Martin’s team at the HHU’s Institute for Molecular Evolution is looking into how and where life emerged on the early Earth. Their method is a combination of experimentation and computation. They use catalysts and conditions found in subsurface hydrothermal vents to conduct chemical experiments in the lab to examine interactions between hydrogen and carbon dioxide, CO2. They’ve devised a computer-based sort of molecular archaeology that allows them to uncover the many distinct remnants of primordial life that are maintained in modern cells’ proteins, DNA, and chemical interactions.
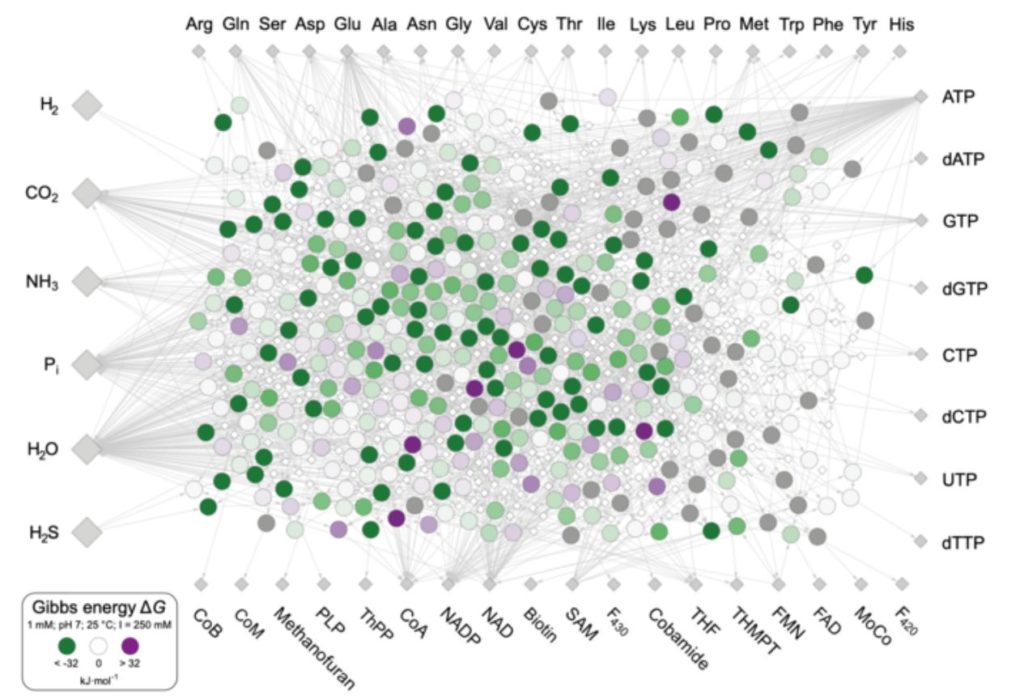
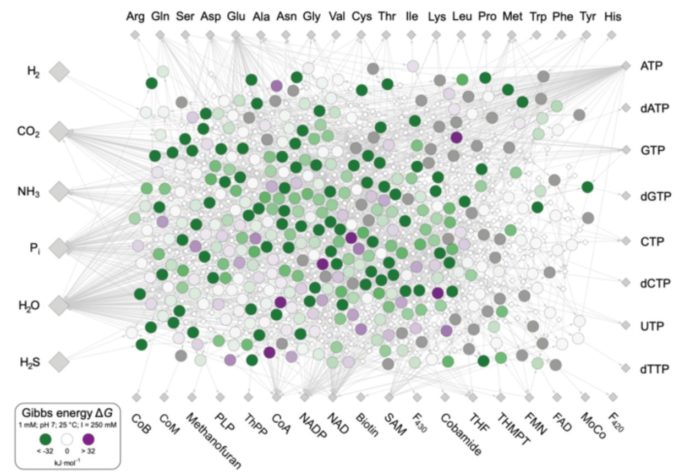
In their most recent research, they looked at what kind of chemical environment favored the chemical processes that gave rise to metabolism and, subsequently, LUCA, as well as where the energy needed to propel those reactions forward came from. They did it by looking at the information inherent inside the chemical interactions of life itself, rather than DNA. They discovered 402 biochemical reactions that have remained nearly unaltered since the beginning of life, about 4 billion years ago. These reactions are found in all cells, hence they were found in LUCA as well. They revealed how primordial life coped with energy in metabolism and where it got the energy it needed to keep life’s chemical interactions moving.
Because all life requires energy, Jessica Wimmer, a PhD student at the institute and primary author on the new work, was particularly interested in the energy balance of LUCA’s metabolic events. She created a list of the 402 processes that bacteria and archaea utilize to generate the building blocks of life: the 20 amino acids, DNA and RNA bases, and the 18 vitamins (cofactors) that are required for metabolism. These substances are created in the most primitive of current cells, as well as in Wimmer’s computer studies, from simple molecules found in today’s environment and in hydrothermal vents on the early Earth: hydrogen (H2), carbon dioxide (CO2), and ammonia (NH3) (NH3). The LUCA metabolic network was born as a result.
Jessica Wimmer, when questioned about the reason for the new study, says: “We wanted to know where the energy came from that drove primordial metabolism forward. At the very onset of metabolic reactions some 4 billion years ago, there were no proteins or enzymes to catalyze reactions because they had not yet evolved. Metabolism had to arise from reactions that could take place in the environment, perhaps with help from inorganic catalysts. But catalysts or not, in order to go forward, the reactions have to release energy. Where did that energy come from? There have been lots of suggestions for possible sources of metabolic energy in the literature. But nobody ever looked into the reactions of metabolism itself.”
The team analyzed the amount of free energy, also known as Gibbs energy, released or consumed in each metabolic reaction to finding energy sources.
As a result, LUCA’s metabolism did not require any external energy sources like as ultraviolet light, meteorite impacts, volcanic eruptions, or radioactivity. In an environment like that of many modern submarine hydrothermal vents, on the other hand, the energy required for metabolism’s reactions to proceed comes from within metabolism itself. To put it another way, practically all of LUCA’s metabolic events release energy on their own: life’s energy comes from life itself.
Martin, the senior author of the study, adds: “That is exciting, because the 400 interconnected reactions of central metabolism, which seem so hopelessly complex upon first encounter, suddenly reveal a natural tendency to unfold all by themselves under the right conditions.”
To reach that result, the researchers had to first look at the energetics of the 402 reactions using computer programs that simulate various environmental conditions in order to discern between energetically favorable and unfavorable combinations. This is significant because whether or not a process releases energy is frequently influenced by external factors. They looked at pH levels ranging from 1 (acidic) to 14 (alkaline), temperatures ranging from 25 to 100 °C, and various ratios of reactants to products. They focused their attention on hydrogen’s energetic role.
Wimmer: “Without hydrogen, nothing happens at all, because hydrogen is required to get carbon from CO2 incorporated into metabolism in the first place.”
With hydrogen necessary for CO2 fixation, the energetically ideal circumstances are an alkaline pH range about pH 9 and a temperature of 80 °C.
Martin explains how this data fits into the bigger picture: “This is almost exactly what we see at Lost City, a H2-producing hydrothermal field in the Mid-Atlantic. In an environment like that, about 95–97 % of LUCA’s metabolic reactions could go forward spontaneously, that is, without the need for any other source of energy. In the abyssal darkness of hydrothermal systems, H2 is chemical sunlight. Modern energy research exploits exactly the same properties of hydrogen as life does. It is just that life has four billion years of experience with hydrogen technology, while we are just getting started.”
Jessica Wimmer further adds: “Regarding energy at life’s origin, we can say that pure chemical energy is sufficient. We need no sunlight, no meteorites, no UV light: just H2 and CO2, plus some ammonia and salts. Because of the extremely conserved nature of the chemical reactions in our biosynthetic network, we can obtain some interesting insights into the reactions that gave rise to LUCA, even though it lived four billion years ago.“
Source: 10.3389/fmicb.2021.793664
Image Credit: HHU / Jessica Wimmer
You were reading: Life’s energy comes from life itself: “We need no sunlight, no meteorites, no UV light”