Scientists at the Russian National University of Science and Technology MISIS (NUST MISIS) together with their colleagues from Russia and Germany have designed an effective substitute for expensive lithium-ion batteries.
According to the researchers, they have discovered a method of manufacturing sodium batteries that will be much cheaper than lithium-ion batteries and may even have a higher capacity. The research results are published in the journal Nano Energy.
Lithium-ion batteries are used today in all areas: in smartphones, laptops, and other electronic equipment, in transportation, and space equipment. Lithium is an expensive metal, and experts say its reserves are limited.
There is currently no effective alternative to these batteries. Lithium is an alkali metal and one of the lightest elements on the periodic table, making it difficult to find a substitution for, according to scientists.
Scientists from NUST MISIS, the Institute of Biochemical Physics under the Russian Academy of Sciences (IBJF) and the Helmholtz Dresden-Rossendorf Center (Germany) led by Professor Arkadi Krasheninnikov have shown a promising alternative to expensive metal. Scientists have shown that by “placing” the alkali metal atoms in a special way, energy capacity comparable to that of lithium can be obtained.
According to the authors, it is proposed to superimpose several layers of atoms. This can be done by letting the metal atoms pass with high tension into a space between two graphene sheets.
“It was believed that only one layer of atoms could be placed in batteries, while recent trials by our colleagues in Germany highlighted that stable multi-layer lithium structures can be created between graphene layers. It was interesting to study the possibility of forming such ‘sandwiches’ with the use of other alkali metals”, highlights one of the authors of the research, an employee of the Inorganic Nanomaterials Laboratory of the NUST MISIS, Ilia Chepkasov.
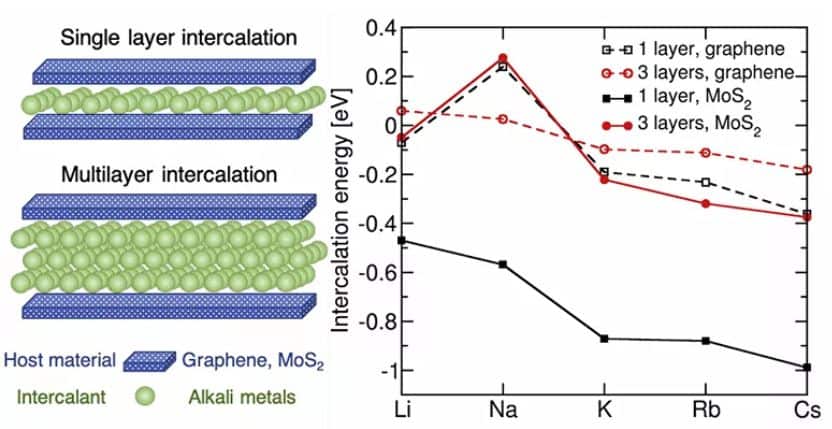
According to scientists, the most promising material that can replace lithium is sodium, which is much more widespread in nature than lithium. For example, table salt contains this element.
According to the model designed by experts, by placing a two-atom thick layer of sodium between two layers of graphene, an anode can be created whose capacity will be slightly lower than that of the anode in lithium-ion batteries: 335 mAh / g compared to 372 mAh / g for lithium.
“The calculations carried out showed that an increase in the number of lithium layers leads to loss of stability. In the case of sodium, the opposite is observed: with the increase in the number of layers, the stability of the structures increases,” explains the employee of the Inorganic Nanomaterials Laboratory of NUST MISIS and IBJF, Zajar Popov.
The next step for the scientific group is to create and study experimental models. According to the scientists, if success is achieved, it will be possible to talk about the creation of a new generation of batteries that will not have a lower capacity than lithium-ion batteries and will even exceed it. The cost of sodium batteries will be considerably less.
