The findings could explain why amyloid-depleting medications haven’t been able to stop the disease.
Alzheimer’s disease affects more than 6 million Americans, the majority of whom are 65 or older, according to the National Institute on Aging. Dementia is a progressive loss of thinking, remembering, and reasoning caused by Alzheimer’s disease.
For decades, scientists have argued that plaques containing the protein amyloid-beta forming outside of cells were a critical initial step toward the brain damage seen in Alzheimer’s disease.
The latest study, led by researchers from NYU Grossman School of Medicine and the Nathan Kline Institute, casts doubt on the amyloid cascade hypothesis.
The latest research suggests that the neuronal damage that characterizes Alzheimer’s disease begins inside cells, long before the thread-like amyloid plaques fully form and cluster together in the brain.
The study, which was published as the cover story in the journal Nature Neuroscience online on June 2, linked the root dysfunction seen in mice bred to develop Alzheimer’s disease to the lysosomes of brain cells. These are tiny sacs inside each cell that contain acidic enzymes that are involved in the ordinary breakdown, elimination, and recycling of metabolic waste generated by everyday cell responses as well as disease. Lysosomes are also important for breaking down and disposing of a cell’s own pieces when it dies naturally, according to researchers.
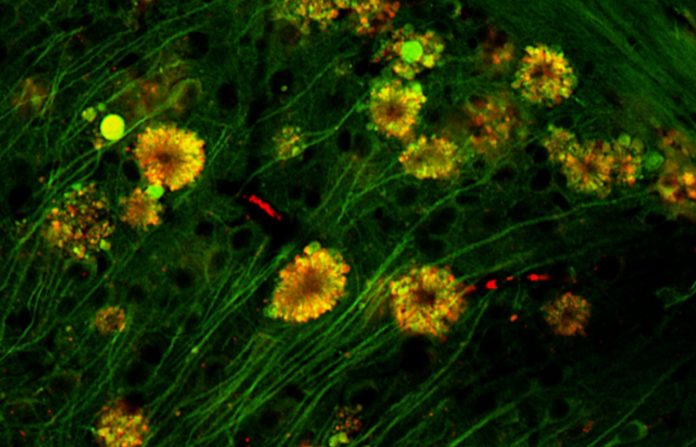
During this investigation, researchers observed a decrease in lysosomal acid activity as the disease progressed. Certain brain cell lysosomes got bigger as they joined with so-called autophagic vacuoles loaded with waste that had failed to be broken down, according to imaging tests developed at NYU Langone Health and Nathan Kline (to track cellular waste elimination). Early forms of amyloid beta were also found in these autophagic vacuoles.
In neurons that had been hurt the most and were going to die soon because of it, the vacuoles clumped together in “flower-like” patterns, bulging out from the outer membranes of the cells and gathering around the center, or nucleus, of each cell. Amyloid beta built up inside the cell to form filaments, which is another sign of Alzheimer’s disease. In fact, some damaged neurons had plaques that were almost completely formed.
“Our results for the first time sources neuronal damage observed in Alzheimer’s disease to problems inside brain cells’ lysosomes where amyloid beta first appears,” explains research lead author Ju-Hyun Lee, PhD.
“Previously, the working hypothesis mostly attributed the damage observed in Alzheimer’s disease to what came after amyloid buildup outside of brain cells, not before and from within neurons,” asserts Lee.
“This new evidence changes our fundamental understanding of how Alzheimer’s disease progresses,” tells study senior investigator Ralph Nixon, MD, PhD. “It also explains why so many experimental therapies designed to remove amyloid plaques have failed to stop disease progression, because the brain cells are already crippled before the plaques fully form outside the cell.”
“Our research suggests that future treatments should focus on reversing the lysosomal dysfunction and rebalancing acid levels inside the brain’s neurons,” notes Nixon.
Researchers said they’re already developing experimental medicines to cure the lysosomal issues they discovered in their research.
A recent study from the NYU Langone team (published in April in Science Advances) linked a gene called PSEN1 to one of the cell’s waste disposal issues. The gene has been linked to Alzheimer’s disease for a long time, but its extra significance in the disease (through lysosomal dysfunction) is only now becoming obvious.
Their latest research also revealed that restoring correct acid levels in lysosomes could restore neuronal damage in a PSEN1 mice model of Alzheimer’s disease.
Image Credit: Springer-Nature Publishing
You were reading: Study Finds A New Target to Treat Alzheimer’s Disease