Scientists from Germany for the first time built a microcapsule from two liquids, which does not collapse during movement. For this, scientists used a nematic liquid crystal as a shell material: in it, the elastic force balances the drag force arising from the circulation of the fluid inside the droplet, and the droplet remains stable. Moreover, the drop spontaneously accelerates, throwing the shell material into the environment.
Scientists have long been developing controlled microscopic devices. In addition to obvious medical applications – for example, for targeted drug delivery to organs – such devices are also needed to create microsensors and microreactors. One of the most common (and simplest) examples of such a device is a microcapsule collected from two immiscible liquids. In fact, such a capsule is a drop-in-drop nucleus surrounded by a spherically symmetrical shell. Over the past ten years, with the help of microcapsules, scientists have learned how to store drugs and food, conduct controlled chemical reactions, collect artificial cell membranes and search certain types of bacteria.
Unfortunately, liquid microcapsules are very easily destroyed: once the core is displaced relative to the centre of the drop, its shell begins to thin. In the end, the shell breaks, and the contents of the droplet flows out. Because of this, the liquid microcapsule is almost impossible to budge. It turns out that such devices are not suitable for the most important practical application – targeted drug delivery – although they have many other advantages.
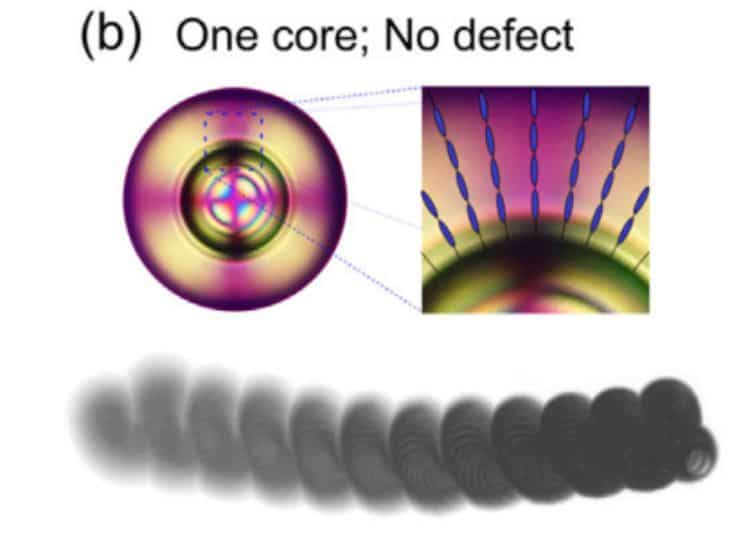
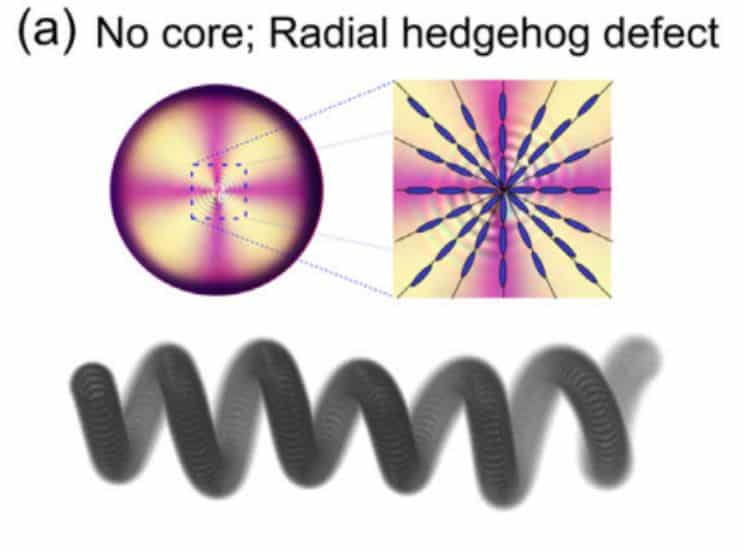
A group of physicists led by Babak Hokmabad corrected this significant flaw. For this, scientists replaced the liquid forming the droplet shell with a nematic liquid crystal. It turned out that due to the violation of symmetry, such a shell creates anisotropic chemical fields and inhomogeneous hydrodynamic forces that keep it in equilibrium. Moreover, physicists have shown that with the help of these forces it is possible to control the motion of a drop, causing it to spontaneously accelerate.
As materials for the capsule, scientists chose water and a nematic liquid crystal 5CB (4-cyano-4′-pentylbiphenyl). To create the droplets, physicists passed the nematic through a narrow tube with two inlet pipes for water: using the first tube, scientists injected a drop into the nematic, and with the second one “cut off” another drop. The characteristic droplet size was several tens of micrometres. Researchers then placed a drop in a surfactant solution (anionic surfactant TTAB). Scientists tracked the movements of droplets in such a solution under a microscope and recorded on video.
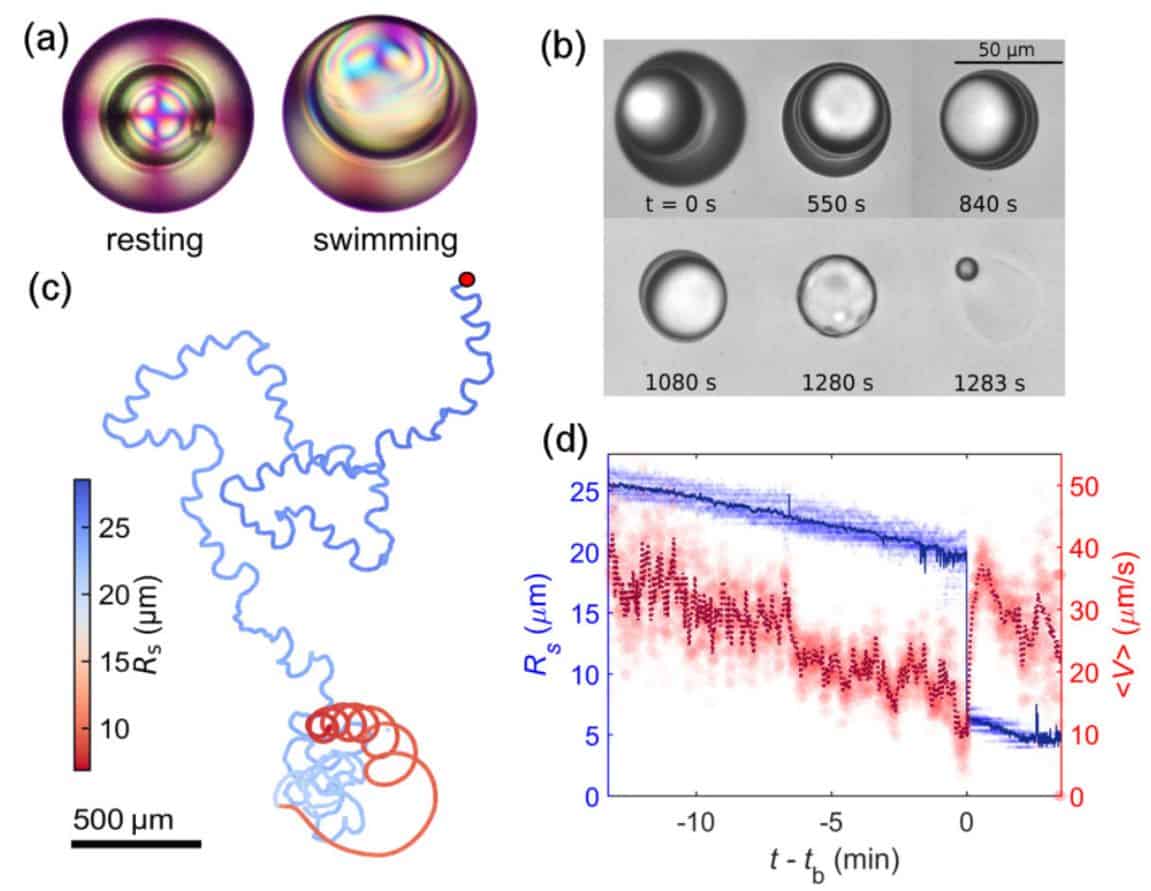
As a result, it turned out that the droplets remain stable for a long time even in those cases when the inner core is strongly shifted relative to the centre of the droplet. At a temperature of about 24 degrees, droplets on average “lived” for about half an hour. Moreover, during this time, the droplets spontaneously accelerated, throwing part of the substance into the environment. In this case, the droplet shell gradually thinned, although it did not completely collapse, and the droplet motion slowed down. The drop fell apart only when the ratio of the radius of the inner core to the thickness of the shell reached twenty. Drops with an isotropic shell under the same conditions disintegrated a hundred times faster, not having time to get to the state of a thin shell. For example, at a temperature of the order of 35 degrees, at which 5CB loses long-range order and ceases to be a liquid crystal.
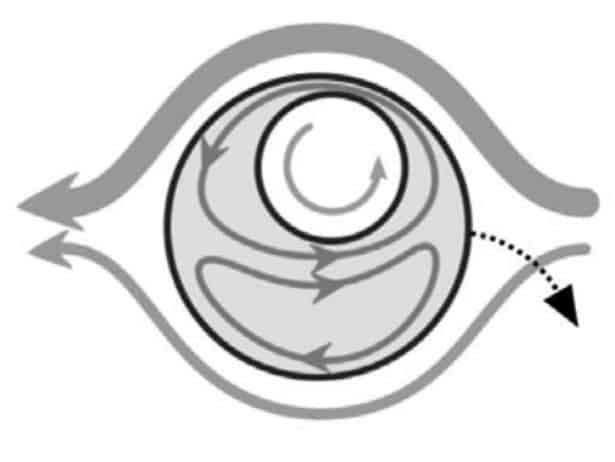
To explain why droplets have remained stable for so long, physicists have numerically modelled the behaviour of the core inside the droplet. On the one hand, fluid is constantly circulating inside the droplet – therefore, the drag force acts on the core, which tries to push it out of the droplet. Scientists have found this power by numerically simulating fluid circulation. On the other hand, the elastic force tends to return it to the centre of the drop. To find this force, scientists calculated the energy of the elastic deformations of the droplet and differentiated it along the radius. As a result, it turned out that near the boundary of the shell, both forces are in order equal to 100 piconewtons and approximately balance each other.
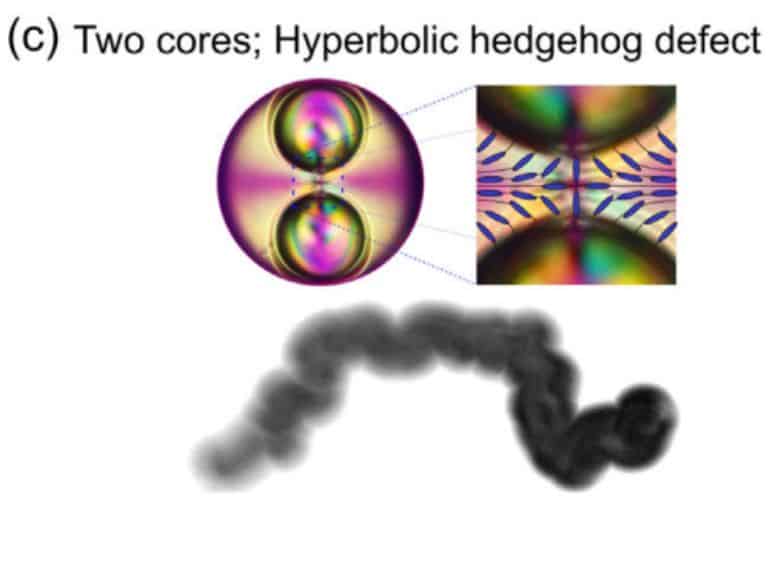
To test this claim, scientists also examined the behaviour of droplets with multiple water cores. Both numerical calculations and the experiment showed that such droplets also remain stable for a long time. However, the extra core has a dramatic effect on how a drop will release shell material into the environment. Therefore, scientists believe that with the help of such drops it is quite possible to deliver drugs or other substances in a targeted manner. Moreover, they can be forced to release matter at a given point by heating them and turning a liquid crystal into a regular isotropic liquid.
Currently, microrobots are rarely used for targeted drug delivery, but scientists are gradually improving this technology.
