Scientists from MIT and Seoul University studied the behaviour of the ammonia molecule in the argon matrix in strong electric fields with up to 200 million volts per meter. It turned out that in fields above 47 million volts per meter, the transitions between the vibrational levels that allowed the molecule to turn out are not observed, so how the outer field guided the molecule and destabilized the turned out state.
The ammonia molecule is a tripod at the top of which is a nitrogen atom. Like an umbrella in the wind, it can turn out, and the nitrogen atom, in this case, is on the other side of the plane, which is hydrogen atoms. The energy barrier that separates these two identical stationary states is quite high (about two thousand reverse centimetres), but the fluctuations are still observed even if the energy is insufficient, as the molecule is able to tunnel through the barrier and move into an inverted state.
To study unstable particles or individual molecules, it is convenient to use the method of matrix isolation. At very low temperatures (kelvin units), the particles studied are isolated from each other by inert gas atoms, and do not interact. Ammonia in such matrix behaves similarly to behaviour in the gas phase, rotates freely, but essentially filled only lower rotational levels.
Youngwook Park and colleagues from Seoul University checked whether ammonia inversion would occur in a field of a strong electric field. To do this, at a temperature of ten kelvins, they placed a film from a mixture of the studied gas in the argon, protected on the both sides by layers of pure argon, between the electrodes, included a constant electric field of up to 200 million volts per meter and recorded absorption and reflection spectrums in the infrared region where it is possible to identify oscillating transitions.

In low temperatures, only one fairly intense spectral line of the desired transition could be observed. With the inclusion of a field of more than 13 million volts per meter on the spectrum, there was another peak, and with a further increase in voltage, the two peaks became closer and at 47 million volts per meter merged into one that became larger and shifted into high-energy areas with rising voltage. By reducing tension, the researchers observed reverse processes.
The action of a strong electric field guided the molecule in the argon matrix across the field, creating a constant electrical dipole moment of the molecule. Interacting with the dipole, the electric field stabilized one state and destabilized another, inverted. The surface of the potential energy became more and more asymmetrical, and at high field values, the molecule was unable to change the conformation.
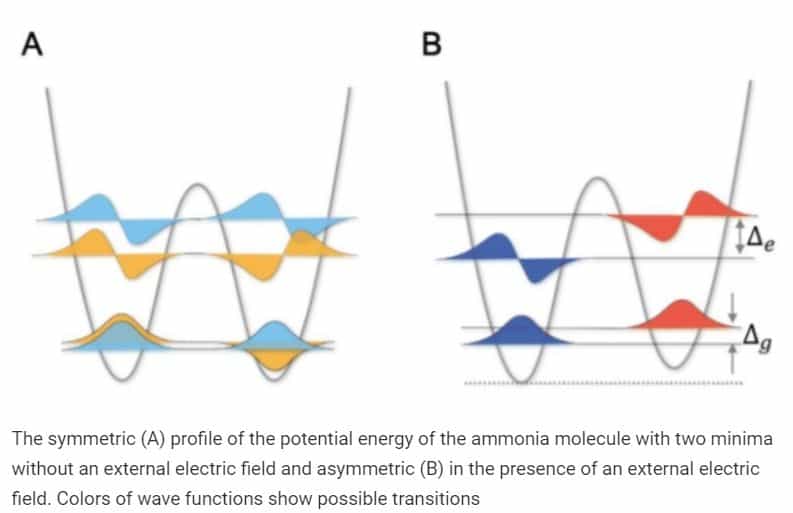
According to the researchers, the new study will bring closer to understanding the effects of electric fields on similar conformational transitions in more complex molecules, and the resulting spectrums ammonia will prove useful for scientists who need to take this influence into account in their experiments.