Physicists from the United States and New Zealand have learned to catch and reverse the quantum leap between the ground and excited states of a three-level artificial atom. For this, scientists followed the signal taken from the auxiliary energy level of the atom.
2019 will be tough for smartphone manufacturers, especially Apple
In 1913, Niels Bohr proposed the famous model of the atomic nucleus, in which electrons move in orbits of a fixed radius and instantly jump between orbits during absorption and emission of photons. For the next seventy years, physicists could not say for sure whether quantum jumps really exist. However, in 1986, three groups of scientists at once independently reported [ 1 , 2 , 3 ] about experimental observation of quantum jumps. All three groups worked with individual barium or mercury ions trapped in a radio frequency trap. Since then, scientists have observed quantum jumps for a number of other ions or solid-state systems.
Windows Hacks: The 44 best tricks of Windows 10
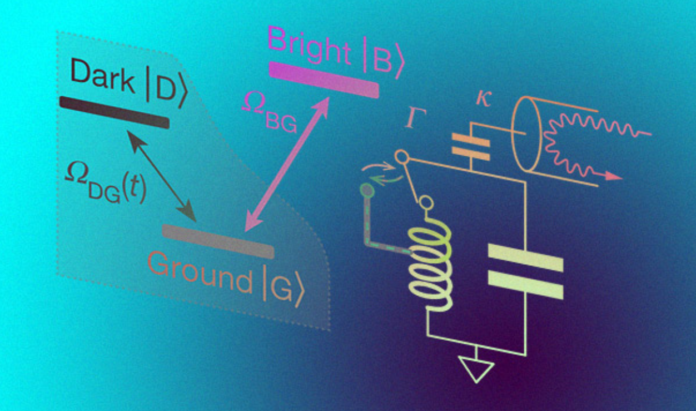
Developed in the late 1980s, the theory of quantum trajectories predicts two unexpected results. First, during the jump, the state of the system evolves continuously over a finite period of time. Secondly, the jump is always preceded by a hidden period during which it can be predicted and prevented by sending the necessary signal to the system. The second result is easiest to explain using the example of a three-level system, which can be mainly | G⟩, excited | D⟩, or an auxiliary | B⟩ quantum state. The | B⟩ state is associated with the detector, the | G⟩ state is hidden from it as best as possible. Suppose the system is constantly excited, so that the state | G⟩ constantly jumps to the state | D⟩ or | B⟩. Since an excited state cannot exist indefinitely by itself, sooner or later the system returns to its ground state. If it was in the | B⟩ state before, the detector will register a signal. If the system was in the state | D⟩, there will be no signal. Therefore, by observing the signal, it is possible (although not absolutely accurate) to say when the system was in an excited or auxiliary state. Moreover, by attenuating the detector signal, one can guess that the system will soon jump to the | D⟩ state. The higher the excitation rate of the | G⟩ state and the decay of the | B⟩ state compared to the decay rate of the | D⟩ state, the more accurately the transition moment can be caught. by weakening the detector signal, one can guess that the system will soon jump to the | D⟩ state. The higher the excitation rate of the | G⟩ state and the decay of the | B⟩ state compared to the decay rate of the | D⟩ state, the more accurately the transition moment can be caught. by weakening the detector signal, one can guess that the system will soon jump to the | D⟩ state. The higher the excitation rate of the | G⟩ state and the decay of the | B⟩ state compared to the decay rate of the | D⟩ state, the more accurately the transition moment can be caught.
Stay locked and unheated: Google’s blackout demonstrates that we are not prepared for the all-connected
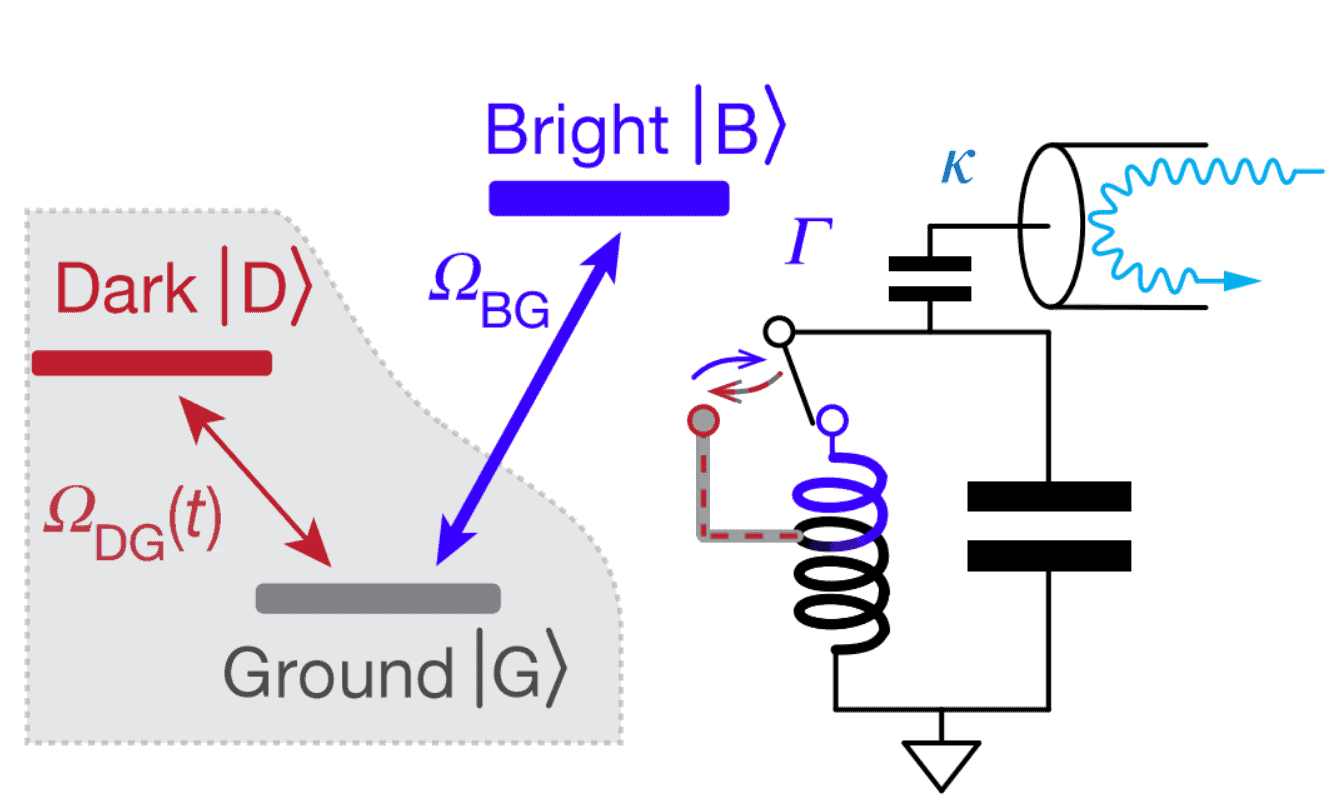
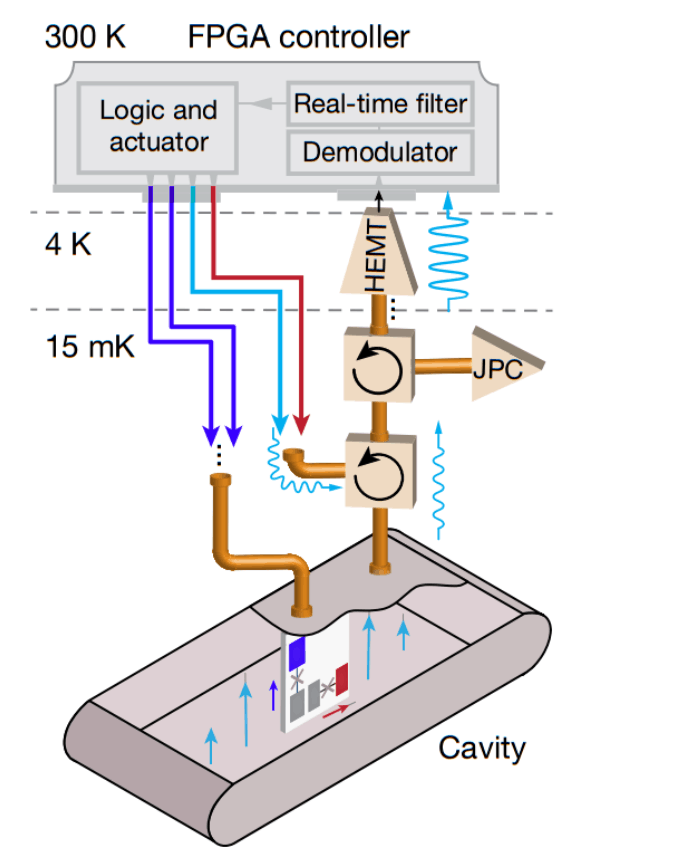
A group of physicists under the leadership of Michel Devoret implemented such a scheme in practice, confirmed the continuity of the quantum jump, learned to predict and prevent it. As a three-level system, scientists used a superconducting artificial atom (qubit) with a V-shaped structure of energy levels, cooled to a temperature of 0.015 kelvins. To read the signal from an artificial atom, the scientists connected the | B⟩ state of the optical cavity and connected it to an oscillatory circuit. When the atom went into the | B⟩ state, the resonant frequency of the circuit was almost halved, while it fell by only three percent. Finally, scientists are constantly excited atom with a laser. Adhering to the tactics described above, scientists determined the state of the atom. To tell whether it is in the | B⟩ state with a certainty of one sigma, on average it took a little less than nine seconds.
The yield in the 10 main crops of the world begins to reduce due to climate change
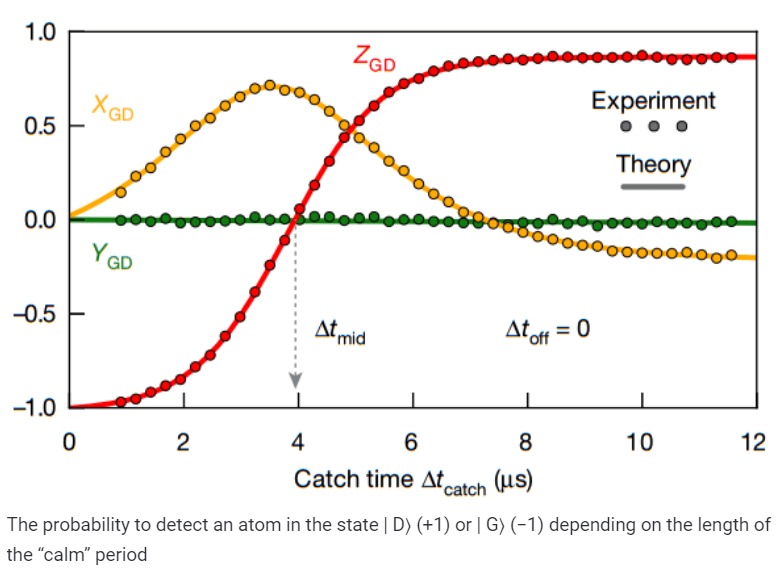
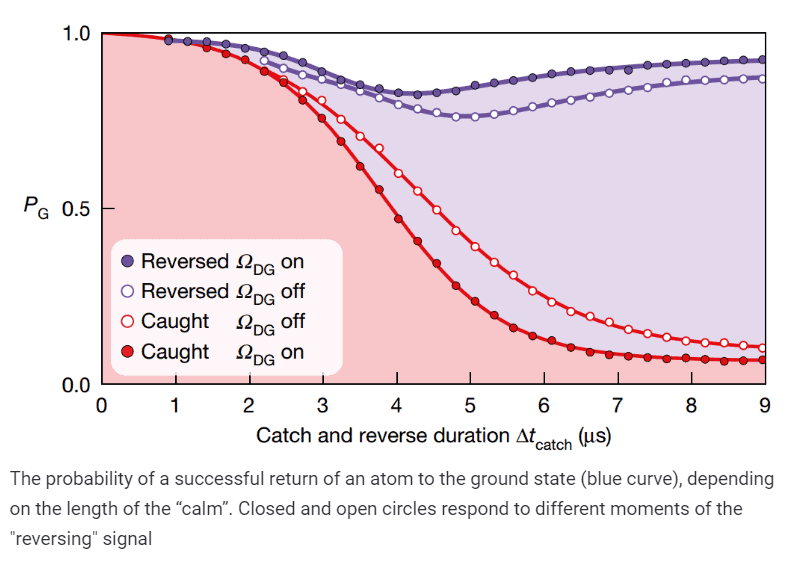
Then the scientists checked that the proposed scheme really allows to capture the moment of transition to the state | D⟩. In other words, if after a “click” of the detector a rather long period of calm followed, the researchers froze the evolution of the system and accurately measured its state using tomography. It turned out that for calm periods shorter than two microseconds, the atom was predominantly in the ground state, and for periods longer than ten microseconds, in the excited state. For intermediate times, the state of the atom changed continuously: despite the fact that each measurement gave only one of two possible results, after averaging over a large number of measurements, scientists obtained a smooth curve. This behavior coincided with the predictions of the theory of quantum trajectories not only qualitatively but also quantitatively.
Market Abuse: Google threatens new antitrust case in the US
Finally, scientists reprogrammed the controller that controlled the excitation of the atom, and forced it to send a reversing signal as soon as there were suspicions about the transition of the atom to the excited state. Thus, physicists managed to reverse the quantum leap back and leave the system in the ground state. Interestingly, the optimal point in time at which the reversing signal had to be applied coincided with half the transition period.
Australian surgeons put out the fire in the patient’s chest
The authors note that the results of their paper do not contradict the probabilistic nature of quantum mechanics. In spite of the fact that in short time intervals, physicists could predict the state of the system, at large times the measurement errors accumulated, and the system behavior again became unpredictable.
USA: Qualcomm convicted of abuse of dominant position
The idea of tracking and controlling the quantum transition using the auxiliary state, on which the Devoret group experiment is based, is not new – at present, similar schemes are already used to correct errors in a quantum computer. In addition, the state of the system can be monitored using an external classical observer. For example, in October 2017, scientists from Germany and Spain with the help of such an observer reversed heat fluxes in a thermionic device, and in January 2018, American physicists learned to monitor the state of a transmon qubit.
First preview of Powershell 7 based on .Net Core 3.0
Source: Nature
How to use WhatsApp on two numbers with the same phone