German Atomic physicists have managed to measure for the first time how long it takes a photon to pass through a hydrogen molecule. This is the shortest time lapse that has been successfully measured to date that is shorter than femtoseconds in magnitude.
They measured the time it takes for a photon to cross a hydrogen molecule – about 247 zeptoseconds, or billionths of a second for the average bond length of the molecule. This is the shortest time period that has been successfully measured to date.
Scientists in Reinhard Dorner’s laboratory carried out time measurement on a hydrogen (H2) molecule that they irradiated with X-rays from the PETRA III X-ray laser source at the DESY acceleration facility in Hamburg.
The researchers set the energy of the X-rays so that one photon was enough to expel both electrons from the hydrogen molecule.
Electrons behave like particles and waves at the same time and, therefore, the release of the first electron resulted in electron waves first launched into the atom of a hydrogen molecule and then the second in rapid succession, merging the waves.
The photon behaved here like a flat stone that slides twice through the water: when a wave channel meets a wave crest, the waves from the first and second contact with the water cancel each other, giving as result what is called an interference pattern.
The scientists measured the interference pattern of the first ejected electron using the COLTRIMS reaction microscope, a device Dörner helped develop that makes ultra-fast reaction processes in atoms and molecules visible.
Simultaneously with the interference pattern, the COLTRIMS reaction microscope also allowed the determination of the orientation of the hydrogen molecule. The researchers took advantage of the fact that the second electron also left the hydrogen molecule, so that the remaining hydrogen nuclei separated and were detected.
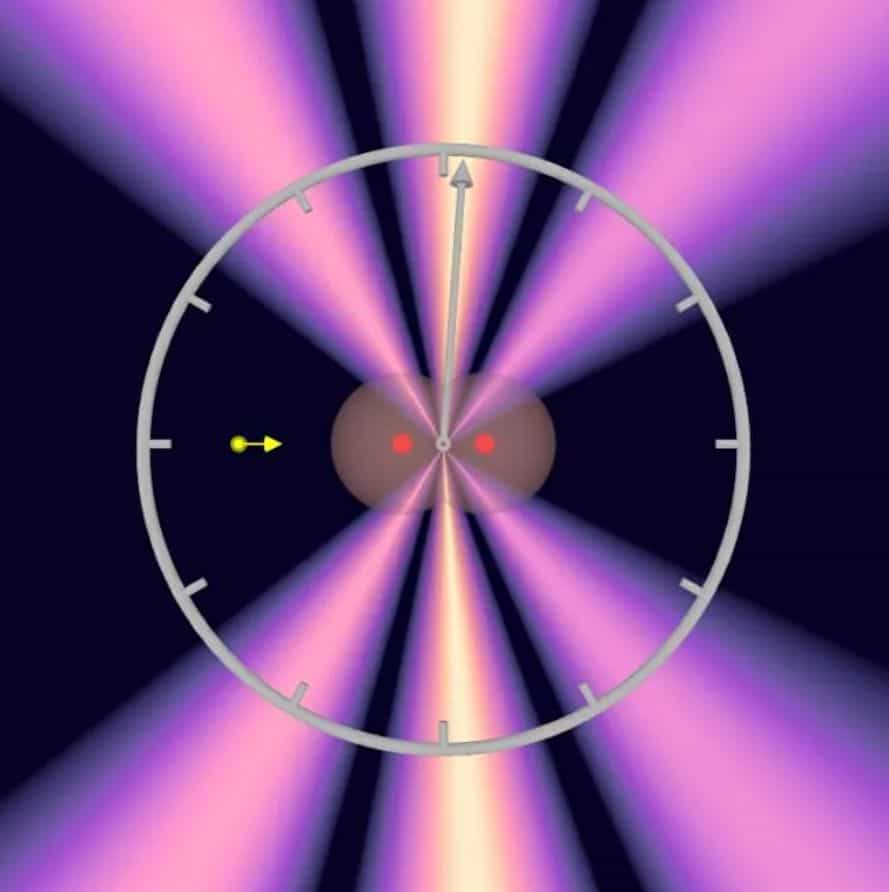
“Since we knew the spatial orientation of the hydrogen molecule, we used the interference of the two electron waves to precisely calculate when the photon reached the first and when it reached the second hydrogen atom,” explains Sven Grundmann, whose doctoral thesis forms the basis of the scientific article published in Science.
“And this is up to 247 zeptoseconds, depending on how far apart in the molecule the two atoms were from the perspective of light.”
Professor Reinhard Dorner added that “We observed for the first time that the electron shell in a molecule does not react to light everywhere at the same time. The time delay occurs because information within the molecule only spreads at the speed of light. With this finding we have extended our COLTRIMS technology to another application.”
