This is an important discovery because it could lead to better vaccines and better ways to treat a wide range of diseases, like infections and autoimmune diseases.
The new study, which was published in the journal Nature Communications, reveals important new information about B cells, which are an important part of the body’s defense system. When we get a vaccine or have an infection, our B cells make antibodies that protect us. However, when we have allergies or autoimmune diseases, our B cells make antibodies that hurt us.
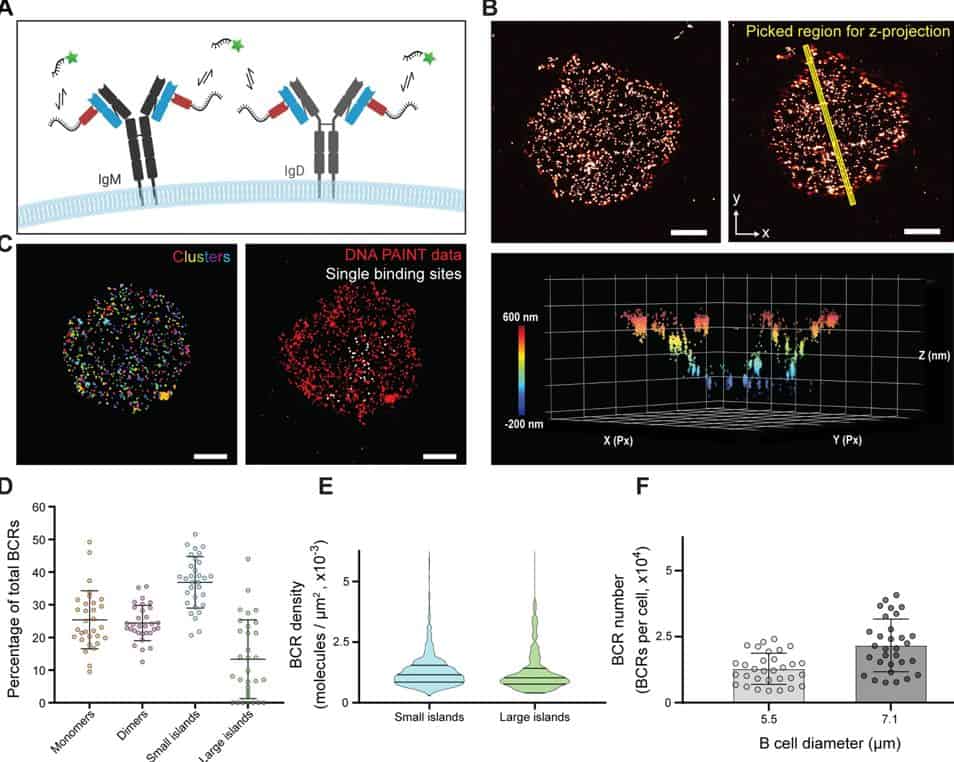
The researchers analyzed the initial phase in B cell activation, specifically the activation mechanism that is activated when the cells detect a particular target or “enemy” — an antigen.
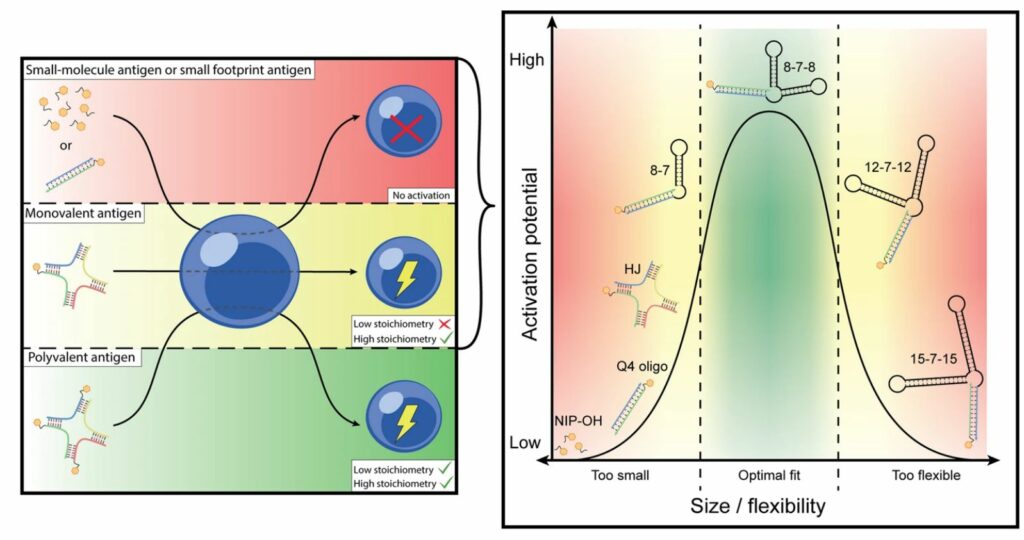
Traditionally, it was thought that for antigens such as those found in viruses or vaccines to activate B-cells, they needed to cross-bind to receptors on the cell surface.
“That’s what it says in all the textbooks,” comments senior author Søren Degn.
However, this research has revealed that even antigens capable of binding only a single receptor at a time can effectively stimulate B-cells.
He says that discovery is important in many ways.
The findings of the study are crucial as “it represents a breakthrough in our understanding of how these important immune cells ‘recognize’ their enemies.”
“Once we understand what is going on,” according to the author, “we can imitate it in the design of new vaccines, to ensure maximum effect.
“One might say that our findings can make us better at mimicking the pathogenic microorganisms, and thus better at provoking or ‘cheating’ the immune system into generating a good immune response when we vaccinate.”
The discovery is of great interest to the fields of immunology and cell biology as it has provided new insights into the fundamental mechanisms underlying how receptors on cell surfaces transmit signals into cells—a critical biological process.
This research “enables us to better understand the background for one of the most important processes in the immune system, and one of the most important processes in cell biology. But it is clear that, in the long term, this could also have important application-oriented aspects,” adds Søren Degn.
The researchers have initiated preclinical vaccination trials in an effort to translate their results into commercially applicable vaccine designs. They are also seeking to reverse-engineer the same tools in order to target and inhibit undesirable immune system responses such as allergy reactions and autoimmune illnesses.
“When we understand how the B cells are activated,” as explained by the author, “we can create better vaccines. In the slightly longer term, we may also be able to switch off B-cell activation in cases where it is harmful. We are studying both of these in the CellPAT basic research center at Aarhus University.”
For a long time, scientists have debated how B cell activation occurs, with the prevailing model for immunological recognition falling short in explaining key facts.
Along with the Max Planck Institute in Munich, researchers from Aarhus’s Department of Biomedicine and iNANO have developed new methods that allow them to put an end to the prevalent model after decades of study.
“We have shown that the way in which the activation of B cells has been explained over the past thirty or forty years is wrong,” comments the author.
“This is an important finding, because it opens the door to better vaccines and better treatment of a large group of diseases.”
Image Credit: Getty