Antibodies kill viruses by latching onto their surfaces and preventing them from infecting host cells, which is well known. However, new research shows that antibodies can destroy viruses in other ways as well.
Antibodies also twist viruses, preventing them from properly adhering to and entering cells, according to an international team of researchers.
“Everybody thinks of antibodies as binding to viruses and blocking them from entering cells — essentially locking them down,” says Ganesh Anand, associate professor of chemistry at Penn State.
“But our research reveals for the first time that antibodies may also physically distort viruses, so they are unable to properly attach to and infect host cells.”
In their research, published today in the journal Cell, Anand and his colleagues analyzed the interactions between the human monoclonal antibody (HMAb) C10 and two disease-causing viruses, Zika and dengue fever. They tested HMAb C10 antibodies that had previously been obtained from dengue virus patients and had been shown to neutralize Zika virus.
The teamr applied a combination of techniques to view the viruses and analyze their movement, including cryogenic electron microscopy (cryo-EM) and hydrogen/deuterium exchange mass spectrometry (HDXMS).
“Cryo-EM involves flash-freezing a solution containing molecules of interest and then targeting them with electrons to generate numerous images of individual molecules in different orientations,” explains the lead author.
“These images are then integrated into one snapshot of what the molecule looks like. The technique provides much more accurate pictures of molecules than other forms of microscopy.”
The scientists used cryo-EM images of the viruses in conditions of increasing antibody concentrations to characterize the effects of antibodies on Zika and dengue viruses.
Simultaneously, the scientists used HDXMS, a technique that involves submerging molecules of interest — in this case, Zika and dengue virus, as well as HMAb C10 antibodies — in heavy water. The hydrogen atoms in heavy water have been replaced with deuterium, hydrogen’s heavier isotope cousin, according to Anand.
“When you submerge a virus in heavy water, the hydrogen atoms on the surface of the virus exchange with deuterium,” the researcher adds.
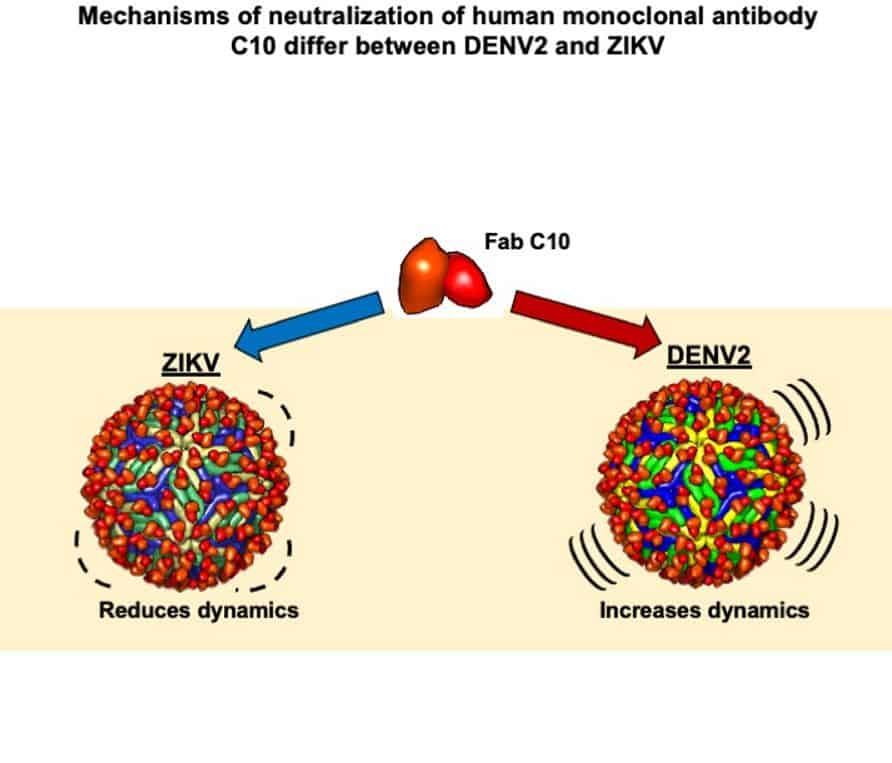
“You can then use mass spectrometry to measure the heaviness of the virus as a function of this deuterium exchange. By doing this, we observed that dengue virus, but not Zika virus, became heavier with deuterium as more antibodies were added to the solution. This suggests that for dengue virus, the antibodies are distorting the virus and allowing more deuterium to get in. It’s as if the virus is getting squished and more surface area becomes exposed where hydrogen can be exchanged for deuterium.”
When Zika virus was placed in heavy water, however, it did not get heavier, implying that its surface, while totally occupied by antibodies, is not deformed by the antibodies.
The team was able to generate a thorough image of what happens when antibodies attach to Zika and dengue viruses by combining cryo-EM with HDXMS, according to Anand.
“It’s like those cartoon flipbooks, where each page has a slightly different image, and when you flip through the book, you see a short movie,” Anand further adds.
“Imagine a flipbook with drawings of a racehorse. Cryo-EM shows you what the racehorse looks like and HDXMS shows you how fast the racehorse is moving. You need both techniques to be able to describe a racehorse in motion. This complementary set of tools enabled us to understand how one type of antibody differentially affects two types of viruses.”
He points out that the more antibodies they added, the more distorted the dengue virus particles became, implying that stoichiometry — the relationship between the amounts of reactants and products before, during, and after a chemical reaction — is important.
“It’s not enough to just have antibodies present. How much antibody you add determines the extent of neutralization.”
In fact, the researchers discovered that when antibodies were introduced in high enough doses to fill all of the accessible binding sites on dengue viruses, 60 percent of the virus surfaces became deformed. This was adequate to keep the cells safe from infection.
“If you have enough antibodies, they will distort the virus particle enough so that it’s preemptively destabilized before it even reaches its target cells.”
Indeed, the scientists discovered that when the antibody-bound dengue viruses were cultured with BHK-21 cells, a cell line derived from the kidneys of young hamsters that is commonly used in viral infection studies, 50-70 percent fewer cells were infected.
Antibodies to some viruses, including Zika, act by jamming the exits, preventing the passenger from exiting the vehicle, according to Anand. In the dengue virus, they discovered a unique method in which antibodies effectively destroy the vehicle, preventing it from even reaching a cell.
How do the antibodies distort the dengue virus particles?
In contrast to the now-familiar SARS-CoV-2, which has spike proteins protruding in all directions, the surfaces of both Zika and dengue are smoother with peaks and valleys, according to Anand.
Antibodies to the dengue virus prefer to bind the ‘peaks’ known as 5-fold vertices, according to the study author. Antibodies will move on to their second-favorite peaks, the 3-fold vertices, once all the proteins on the 5-fold vertices have been bound. They’re left with simply the 2-fold vertices at the end.
“Antibodies do not like two-fold vertices because they are very mobile and difficult to bind to,” adds Anand. “We found that once the 5- and 3-fold vertices have been fully bound with antibodies, if we add more antibodies to the solution, the virus starts to shudder. There’s this competition taking place between antibodies trying to get in and the virus trying to shake them off. As a result, these antibodies end up burrowing into the virus rather than binding onto the 2-fold vertices, and we think it’s this digging into the virus particle that causes the virus to shake and distort and ultimately become non-functional.”
What is the difference between Zika and dengue virus?
Zika is a much more stable, less dynamic virus than dengue, which has a lot of moving elements, says Anand.
“Dengue and Zika look similar but each one has a different give. Dengue may have evolved as a more mobile virus as a way of avoiding being caught by antibodies. Its moving parts confuse and throw off the immune system. Unfortunately for dengue, antibodies have evolved a way around this by burrowing into the virus and distorting it.”
The same sort of antibody can neutralize Zika and dengue in two ways: one by binding to the virus and deactivating it, which is how we usually conceive of antibody activity, and the other by burrowing in and distorting the virus.
What about other viruses?
Anand stated that the distortion approach developed by his team may be adopted by antibodies when they encounter various types of viruses.
“Dengue is just a model virus that we used in our experiments, but we think this preemptive destabilization strategy may be broadly applicable to any virus.
“It may be that the antibodies first attempt to neutralize viruses through the barrier method and if they are unsuccessful, they resort to the distortion method.”
Are there any potential applications of the findings?
The research could allow the development of therapeutic antibodies, the study authors highlight.
“HMAb C10 antibodies are specific to dengue and Zika viruses, and happen to be capable of neutralizing Zika and dengue viruses in two different ways.”
“But you could potentially design therapeutics with the same capabilities for treating other diseases, such as COVID-19. By creating a therapeutic with antibodies that can both block and distort viruses, we can possibly achieve greater neutralization.”
He further adds:
“You don’t want to wait for a virus to reach its target tissue, so if you can introduce such a therapeutic cocktail as a nasal spray where the virus first enters the body, you can prevent it from even entering the system. By doing this, you may even be able to use less antibody since our research shows that it takes less antibody to neutralize a virus through the distortion method. You can get better bang for the buck.”
In general, Anand says that the discovery is significant because it elucidates a previously unknown strategy by which some antibodies block certain viruses.
“Previously, all we knew about antibodies was that they bind and neutralize viruses.
“Now we know that antibodies can neutralize viruses in at least two different ways, and perhaps even more. This research opens the door to a whole new avenue of exploration.”
Image Credit: iStock
You were reading: Antibodies can also distort and disable viruses without latching, blocking them from infecting host cells
