The test could assess patient immunity to multiple COVID-19 variants, such as Omicron and Delta in less than an hour—possibly as little as 15 minutes and also suggest treatment options.
The test could assess patient immunity to several COVID-19 variants, such as Omicron and Delta, at the same time and advise on which synthetic monoclonal antibody to administer for treatment.
Duke University biomedical engineers have developed a test to swiftly and simply determine how successfully a person’s neutralizing antibodies fight infection from several COVID-19 variants, including the recently found Omicron type.
Potentially, this test could tell doctors whether a patient is protected from new COVID-19 variants or which monoclonal antibodies should be used to treat a COVID-19 patient.
“We currently really have no rapid way of assessing variants, neither their presence in an individual, nor the ability of antibodies we possess to make a difference,” says Cameron Wolfe.
“It’s one of the lingering fears that, as we successfully vaccinate more and more people, a variant may emerge that more radically evades vaccine-induced antibody neutralization. And if that fear came true – if Omicron turned out to be a worst-case scenario – how would we know quickly enough?”
“While developing a point-of-care test for COVID-19 antibodies and biomarkers, we realized there could be some benefit to being able to detect the ability of antibodies to neutralize specific variants, so we built a test around that idea,” adds Ashutosh Chilkoti.
“It only took us a week or two to incorporate the Delta variant in our test, and it could easily be expanded to also include the Omicron variant. All we need is the spike protein of this variant, which many groups across the world — including our group at Duke— are feverishly working to produce.”
CoVariant-SCAN, or COVID-19 Variant Spike-ACE2-Competitive Antibody Neutralization assay, is the name given to the test by the researchers. In order to ensure that only the targeted biomarkers adhere to the test slide while wet, the test relies on a polymer brush coating that acts as a form of non-stick coating.
The non-stick shield’s exceptional efficacy makes the test extremely sensitive to even the tiniest levels of its targets. Researchers can use the method to print many molecular traps on the slide at the same time, allowing them to catch various biomarkers.
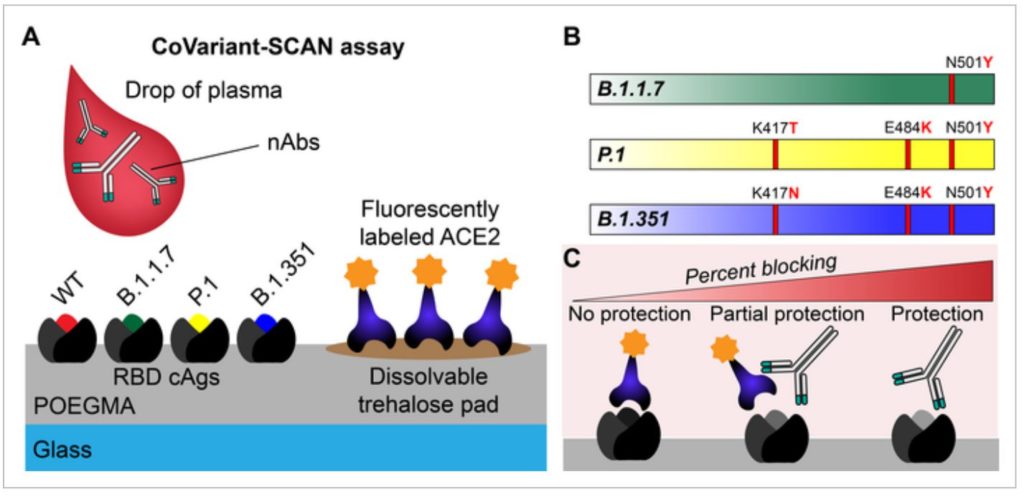
The researchers used this program to print fluorescent human ACE2 proteins on a slide. These proteins are the cellular targets of the virus’s famed spike protein. Additionally, they print spike proteins specific to each COVID-19 variant at distinct sites. When the test is performed, the ACE2 proteins detach off the slide and are captured by the remaining spike proteins, causing the slide to glow.
However, in the presence of neutralizing antibodies, the spike proteins are unable to attach to the ACE2 proteins, causing the slide to light less brightly, showing the antibodies’ effectiveness. By printing several variants of the COVID-19 spike protein on separate regions of the slide, researchers can assess the antibodies’ efficacy in blocking each variant from latching onto its human biological target concurrently.
The researchers evaluated the technology in a variety of ways in the study. They tested monoclonal antibodies generated from real-world patients as well as those from Regeneron’s commercial preventive treatment. Additionally, they analyzed plasma from healthy vaccinated individuals and those who are currently afflicted with the virus.
“In all of our tests, the results largely mimicked what we’ve been seeing in the literature,” says Jake Heggestad.
“And in this case, not finding anything new is a good sign, because it means our test is working just as well as the methods currently being used.”
While both approaches generate comparable results, the fundamental distinction between the CoVariant-SCAN and current methods is the speed and ease with which it produces them. Typical current procedures entail isolating and culturing live virus, which can take up to 24 hours and requires a range of safety precautions and particularly trained specialists. In comparison, the CoVariant-SCAN does not require live virus, is simple to use in most circumstances, and produces accurate results in less than an hour—possibly as little as 15 minutes.
Heggestad and the Chilkoti laboratory are now attempting to simplify the procedure into a microfluidic chip that could be mass produced and report results using only a few drops of blood, plasma, or another liquid sample containing antibodies. This approach has already been demonstrated to be effective in the context of a similar test capable of differentiating COVID-19 from other coronaviruses.
“We would love to have real-time visibility of the emerging variants and understand who still has functional immunity,” adds Wolfe.
“Additionally, this hints that there might be a technique whereby you could quickly assess which synthetic monoclonal antibody might be best to administer to a patient with a particular emergent variant. Currently we really have no real-time way of knowing that, so we rely on epidemiological data that can track weeks behind.”
“The reverse is also true,” Wolfe continues, adding, “to be able to pre-screen an individual’s antibodies and predict whether they were sufficiently protected against a particular variant they are perhaps about to run into while travelling, or that is emerging in their area. We have no way of doing that at the present time. But a test like the CoVariant-SCAN could make all of these scenarios possible.”
Source: 10.1126/sciadv.abl7682
Image Credit: Jake Heggestad, Duke University
You were reading: New Rapid Test Detects Escape of Multiple COVID-19 Variants such as Omicron, Delta