Using these mucins, a new study says, could help researchers come up with new antifungal drugs or make disease-causing fungi more susceptible to drugs they have become resistant to.
Candida albicans is a yeast that can be found in the human gastrointestinal tract, mouth, urinary tract, and reproductive organs. It usually does not cause disease in its host, but under specific circumstances, it can transform into a dangerous version. The majority of Candida infections aren’t fatal, but systemic Candida infections, which affect the blood, heart, and other organs, can be fatal.
Now, scientists at MIT have found parts of mucus that can interact with Candida albicans and stop it from causing infections. These molecules, referred to as glycans, are a major component of mucins, the gel-forming polymers that comprise mucus.
Mucins are made up of many different sugar molecules called glycans. According to Katharina Ribbeck, the Andrew and Erna Viterbi Professor at MIT, a growing body of evidence suggests that glycans might be specialized to assist in tame specific infections — not only Candida albicans but also other pathogens such as Pseudomonas aeruginosa and Staphylococcus aureus.
“The picture that is emerging is that mucus displays an extensive small-molecule library with lots of virulence inhibitors against all sorts of problematic pathogens, ready to be discovered and leveraged,” adds Ribbeck.
Using these mucins could aid researchers in the development of new antifungal treatments or make disease-causing fungus more vulnerable to existing drugs. There are few such medications on the market right now, and some harmful fungi have acquired resistance to them.
Julie Takagi, an MIT graduate student, is the lead author of the work, which was published today in Nature Chemical Biology.
We have fungus among us
Mucus, far from being an inert waste product, plays an active role in keeping potentially hazardous microorganisms under control, as Ribbeck and others have discovered over the last decade. Inside the mucus that covers most of the body, there are communities of different microbes, some of which are good and some of which are bad.
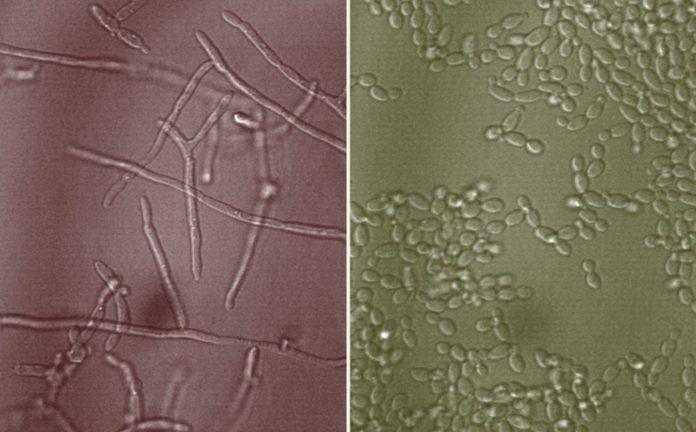
Candida albicans is one of the bacteria that can be dangerous if not controlled, producing thrush and vaginal yeast infections in the mouth and throat. Antifungal medications may usually clear up those infections, but invasive Candida albicans infections of the bloodstream or internal organs, which can occur in persons with weaker immune systems, have a 40 percent death risk.
Mucins can prevent Candida albicans cells from converting from their spherical yeast form to multicellular threads called hyphae, which is the microbe’s dangerous variant, according to Ribbeck’s earlier research. Hyphae can emit poisons that harm the immune system and underlying tissue, and they’re also necessary for the production of biofilms, which are a hallmark of infection.
“Most Candida infections result from pathogenic biofilms, which are intrinsically resistant to the host immune system and antifungal therapeutics, posing significant clinical challenges for treatment,” explains Takagi.
Yeast cells continue to grow and flourish in mucus, but they are not pathogenic.
“These pathogens don’t seem to cause harm in healthy individuals,” Ribbeck explains. “There is something in mucus that has evolved over millions of years, that seems to keep pathogens in check.”
Mucins are a bottlebrush-like structure made up of hundreds of glycans connected to a lengthy protein backbone. Ribbeck and her colleagues wanted to see if glycans could disarm Candida albicans on their own, without the help of the mucin backbone, or if the full mucin molecule was required.
After removing glycans from the backbone, the authors investigated them to Candida albicans and discovered that these glycan collections inhibited the formation of filaments by Candida. They may also inhibit adhesion and biofilm development, as well as change the dynamics of Candida albicans’ interactions with other microorganisms. Mucin glycans from human saliva and animal gastric and intestinal mucus were shown to be similar.
Because isolating single glycans from these collections is difficult, Rachel Hevey’s team at the University of Basel synthesized six different glycans found on mucosal surfaces and used them to see if individual glycans could disarm Candida albicans.
“Individual glycans are nearly impossible to isolate from mucus samples with current technologies,” Hevey adds. “The only way to study the characteristics of individual glycans is to synthesize them, which involves extremely complicated and lengthy chemical procedures.”
There are only a handful of research groups in the world that are developing techniques to synthesis these complicated compounds.
In Ribbeck’s lab, researchers discovered that each of these glycans had at least some potential to inhibit filamentation on their own and that some of them were much more potent than the collections of numerous glycans they had previously studied.
More than 500 genes are upregulated or downregulated in response to interactions with glycans, according to a study on Candida gene expression. These genes included not only filament and biofilm formation genes, but also genes involved in amino acid synthesis and other metabolic processes. Many of these genes appear to be regulated by the transcription factor NRG1, a master regulator triggered by glycans.
“The glycans appear to rewire those microorganisms by tapping into physiological circuits,” Ribbeck explains. “It’s a massive arsenal of chemicals that promote host compatibility,” says the researcher.
The researchers were also able to match individual mucin samples to the glycan structures discovered inside them, allowing them to further investigate how those patterns correspond with microbial behaviour, according to professor Micheal Tiemeyer.
“Using state-of-the-art glycomic methods, we have begun to comprehensively define the richness of mucin glycan diversity and to annotate that diversity into motifs that have functional implications for both host and microbe,” he continues.
A collection of molecules
This research, along with Ribbeck’s past work on Pseudomonas aeruginosa and ongoing research on Staphylococcus aureus and Vibrio cholerae, suggests that distinct glycans are designed to disable different types of microorganisms.
She thinks that by utilizing this diverse set of glycans, researchers may be able to design new treatments for a number of viral disorders. By breaking up the filaments that develop in the pathogenic state, glycans could be used to either stop a Candida infection or assist sensitize it to existing antifungal medicines.
“The glycans alone can potentially reverse an infection, and convert Candida to a growth state that is less harmful to the body,” adds Ribbeck. “They also might sensitize the microbes to antifungals, because they individualize them, thereby also making them more manageable by immune cells.”
Ribbeck is currently collaborating with medicine delivery experts to figure out how to get mucin glycans into the body or onto surfaces like the skin. She’s also working on a number of experiments to see how glycans affect a range of bacteria.
“I am really excited about this new work because I think it has important implications for how we develop new antimicrobial therapies in the future,” says Clarissa Nobile, an associate professor of molecular and cell biology. “If we figure out how to therapeutically deliver or increase these protective mucin glycans into the human mucosal layer, we could potentially prevent and treat infections in humans by maintaining microorganisms in their commensal forms.”
Image Credit: Getty
You were reading: Sugar Molecules In Mucus Can Help Develop New Antifungal Drugs